
29 Mar Find Clinical Trials near you with new Interactive Map
The European Medicines Agency (EMA) has recently launched a new online tool to help patients and healthcare professionals easily find ongoing clinical trials in the European Union (EU). This tool, an interactive clinical trial map, is part of a larger effort to make research more accessible to patients and to encourage participation in clinical studies.
.
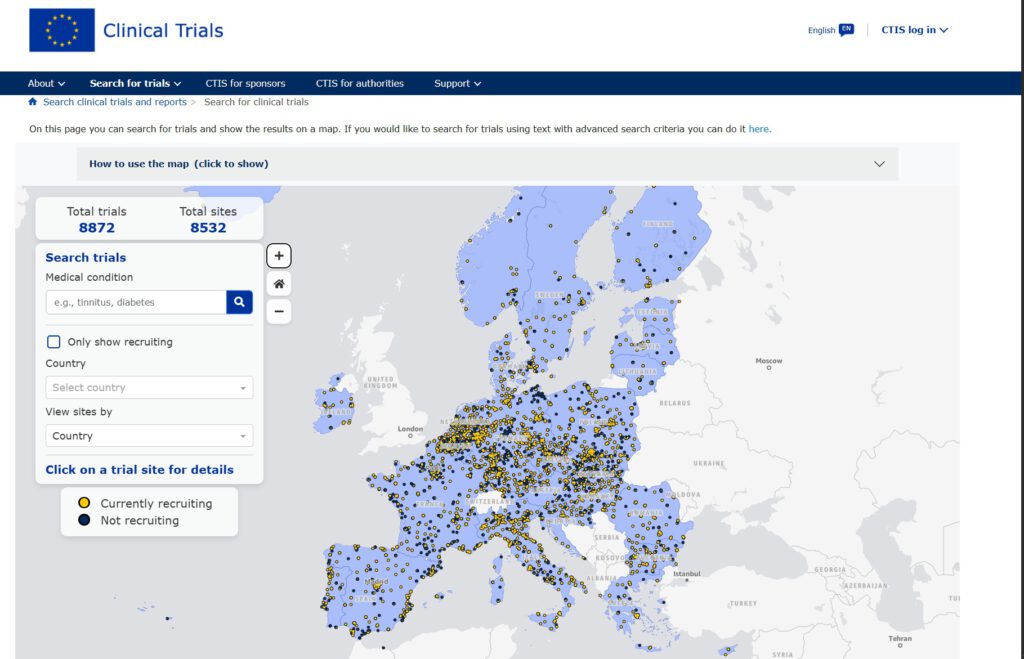
It currently list almost 9,000 trials, which take place in more than 8,500 locations all over the EU.
They are all regulated under the EU Clinical Trials Regulation.
What is the Clinical Trial Map?
The map is available on the Clinical Trials Information System (CTIS) public website. It allows users to search for clinical trials by medical condition or location. One doesn’t need to know complicated medical terms to use it—the tool can recognize simple words and even correct typos. When one finds a clinical trial that is of interest, the map provides contact details for the research team running the trial, so people can further ask questions or explore how to join.
Why is this Important?
Clinical trials are essential to developing new medicines and treatments. They involve testing new drugs, medical devices, or procedures on volunteers to ensure they are safe and effective. By participating, patients not only access potentially life-saving treatments but also contribute to advancing medical science. However, finding information about these trials has often been complicated—this map aims to change that.
How Can You Use It?
The first version of the map is currently available in English, but future updates will add more EU languages.
This map is a game-changer for patients and families who want to explore new treatment options. It’s also a valuable tool for healthcare professionals and researchers seeking to connect with clinical studies.
 The tool is part of the EMA’s broader plan to improve how clinical trials are conducted in the EU. This is outlined in the “Accelerating Clinical Trials in the EU” (ACT EU) workplan for 2025-2026.
The tool is part of the EMA’s broader plan to improve how clinical trials are conducted in the EU. This is outlined in the “Accelerating Clinical Trials in the EU” (ACT EU) workplan for 2025-2026.
Clinical trials included in the map are regulated under the EU Clinical Trials Regulation. This ensures they meet high ethical and safety standards.
The CTIS, introduced in 2022, centralizes all clinical trial data for the EU and European Economic Area (EEA). This platform promotes transparency by publishing information such as trial objectives, design, and outcomes.
For more details, visit the EMA’s official page on the clinical trial map.
